Inflazome was co-founded in 2016 by globally recognised academic scientists, Prof Matt Cooper (University of Queensland) and Prof Luke O’Neill (Trinity College Dublin) following a highly productive collaboration.
The Science
The body's immune system monitors infection or damage to our bodies. It uses special sensors to detect microbes or products of damaged tissues. Inflammasomes are an important family of such sensors. Inflammation normally leads to repair to damaged tissue. However, if inflammation becomes chronic, all kinds of inflammatory diseases can occur.
When immune cells recognise an infection or harmful substance in the body that needs to be neutralised, inflammasomes – groups of proteins in the immune cells – generate signals that cause inflammation and call other cells to help. This short term, beneficial inflammation allows the cells to fight and clear the infection or foreign particles, restoring us to good health.
Inflammation can become harmful if immune cells are activated out of control. Inflammasomes generate signals calling for reinforcements whenever immune cells are stimulated. Sometimes the cells are over-stimulated, and in this case, the resulting overactive inflammatory response can damage healthy tissue, which in turn causes more inflammasomes to trigger, creating more inflammation.
We block inflammasome signals, eliminating the root cause of inflammation. Our small-molecule drugs – drugs that can be taken by mouth – block inflammasomes very precisely. By carefully targeting solely the inflammasome, our therapies have the potential to stop harmful inflammation while allowing beneficial inflammation and minimising side effects.
NLRP3 is the best validated target in inflammation that I have seen in 30 years. At Inflazome we are leading the way in developing highly selective inhibitors which will have tremendous clinical utility for major diseases of our time.
CSO and Co-Founder

Inflammation Cycle

Cells Stimulated
Immune cells are stimulated by noxious inflammatory material such as crystals or protein aggregates.

Healthy Tissue Damaged
Healthy Tissue is damaged by the inflammatory processes.

Generate Signals
Signals are generated via NLRP3 to further provoke inflammation

Cells Gather and Cause Inflammation
Inflammatory cells such as neutrophils and macrophages gather to accelerate tissue injury promoting disease
The Research
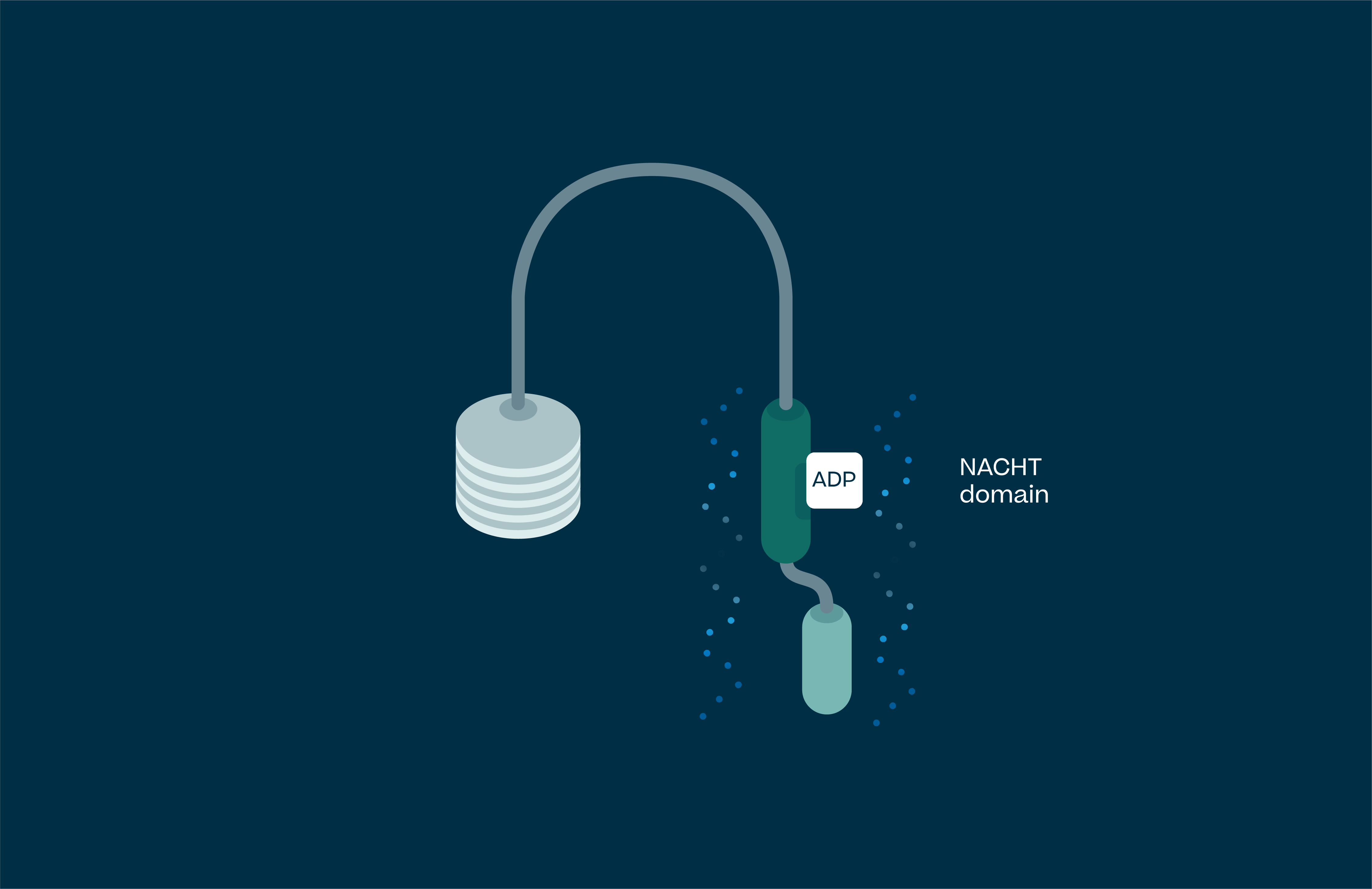
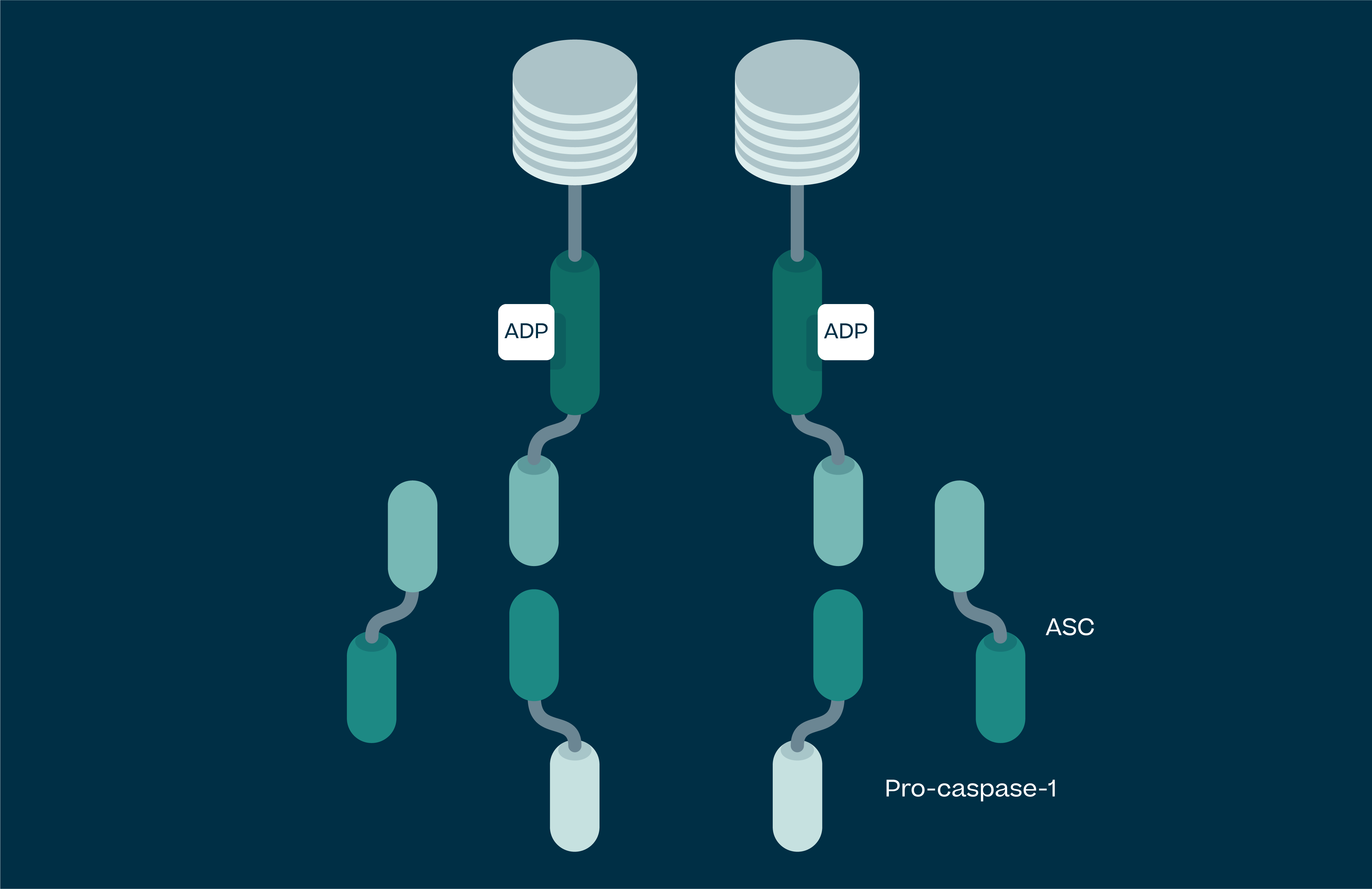
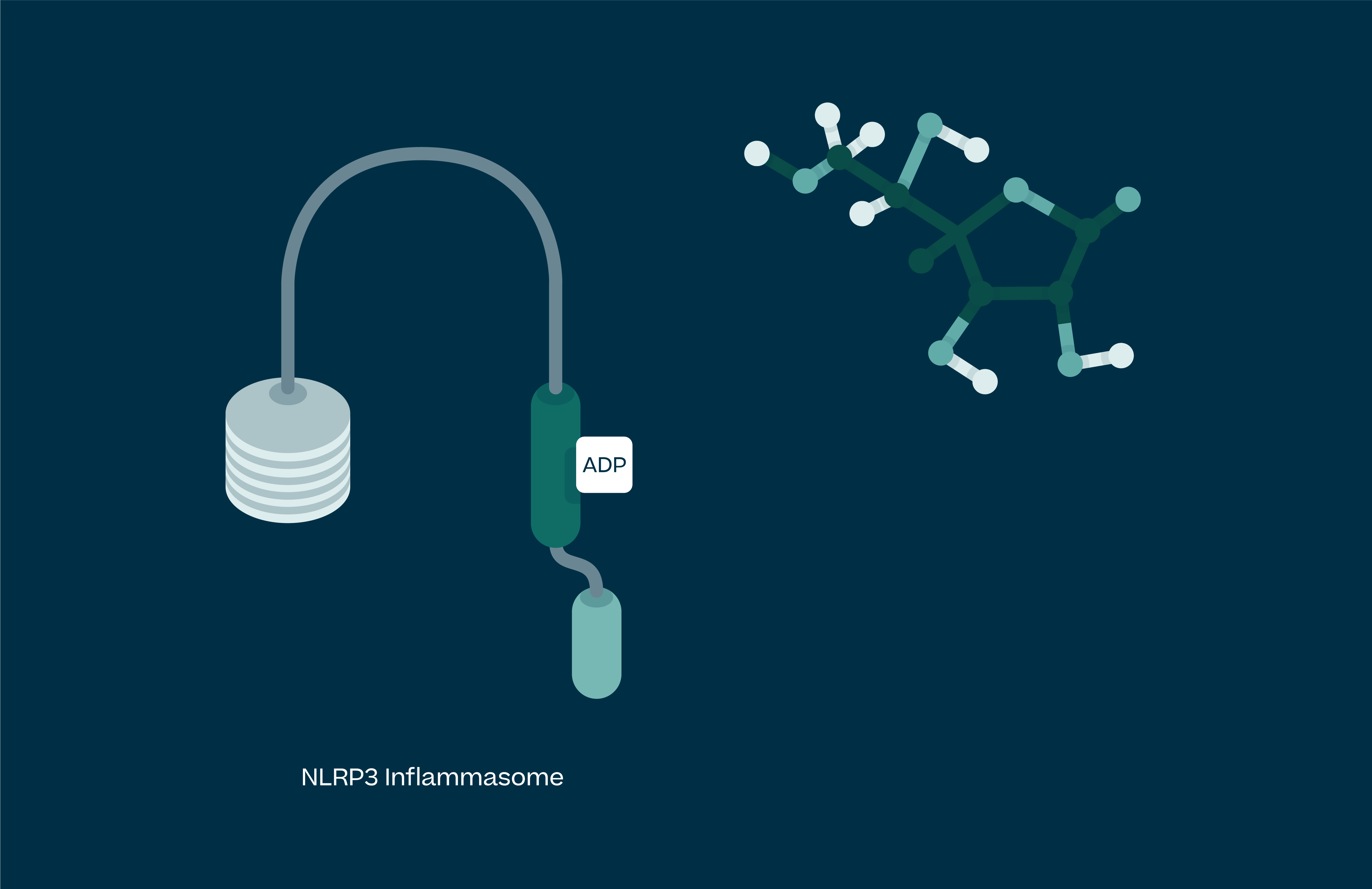
Understanding NLRP3
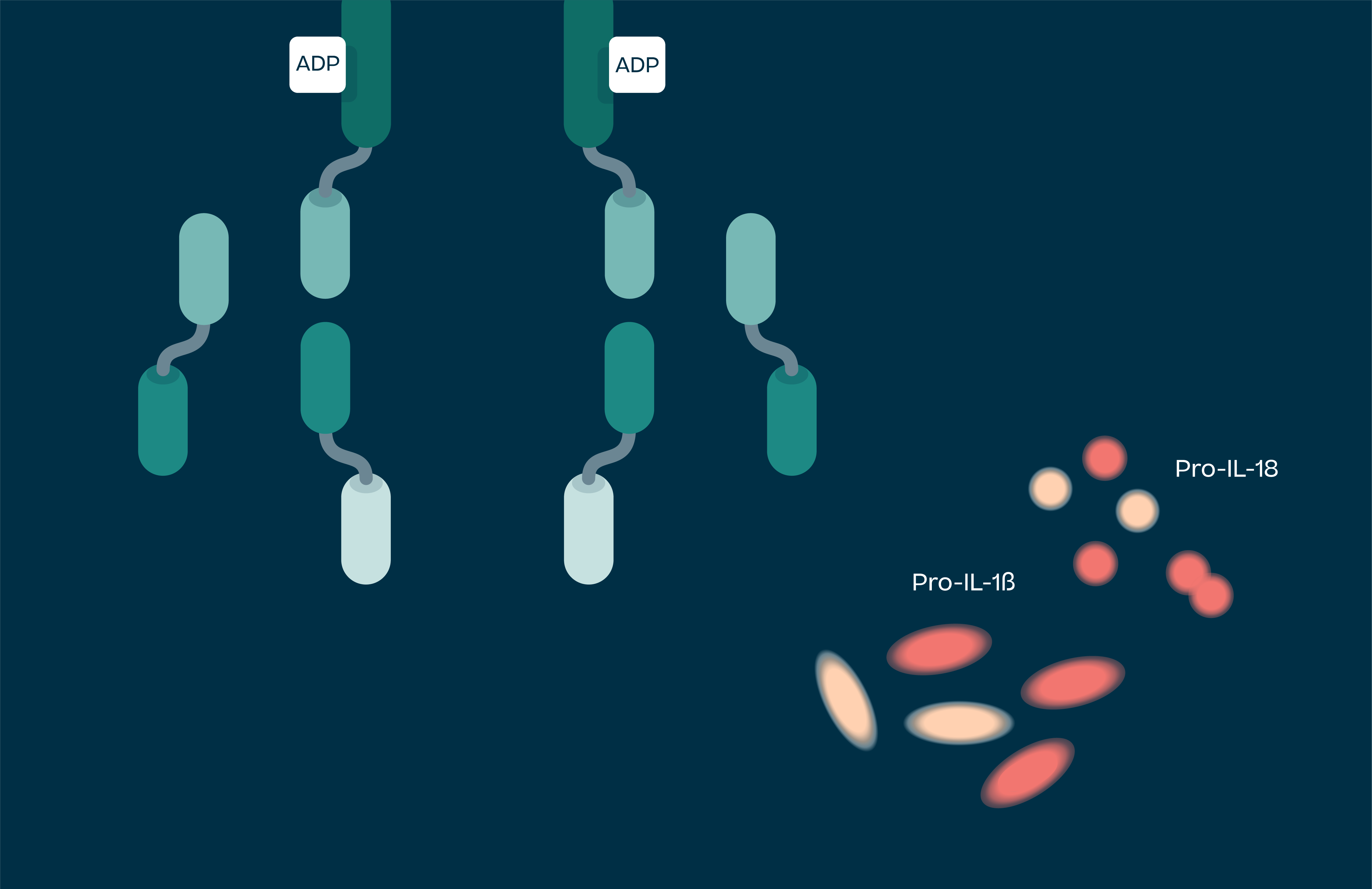
Activated NLRP3 drives the release of pro-inflammatory cytokines IL-1β, IL-18 and uncontrolled cell death (pyroptosis). These processes lead to chronic inflammation in humans, and as such NLRP3 has been implicated in a large number of diseases. These range from autoimmune conditions, the simplest of which is NLRP3 gain of function mutations leading to a syndrome called Cryopyrin Associated Periodic Syndrome (CAPS), to large disease burden indications such as Parkinson’s, Alzheimer’s, atherosclerosis, heart failure, stroke, IBD and NASH.
Is caused by factors external to the cell or tissue, such as infection, toxins, or trauma which result in the unregulated digestion of cell components.
NLRP3 drives release of the pro-inflammatory cytokines IL-1β, IL-18 and pyroptosis (uncontrolled cell death). These processes lead to chronic inflammation in humans, and as such NLRP3 has been implicated in a large number of diseases.
Following Phase I, Inzomelid will be evaluated for proof of concept in patients with gain-of-function NLRP3 mutations that result in the spectrum of diseases collectively termed CAPS (Cryopyrin Associated Periodic Syndrome).
Clinical and scientific data suggests that inflammation causes many of our most challenging medical conditions. By blocking the inflammasome we can stop inflammation from progressing and inflammation-related diseases may never even develop.
At Inflazome we are focussed on developing proprietary, best in class and orally available drugs that block activation of the NLRP3 inflammasome in order to address clinical unmet needs in inflammatory diseases.
Inflazome has a leading IP position in the NLRP3 inhibitor space. Inflazome has produced an extensive patent portfolio to cover our lead clinical candidates, Inzomelid and Somalix. We have also filed patent applications covering potential next generation candidates and the surrounding generic scope to thoroughly cover the chemical matter in this competitive area. The first patent applications were filed in early 2015.
NLRP3 and Disease:
Since the identification of the NLRP3 inflammasome in 2001, many activators of NLRP3 have been identified and associated with diseases. A list of diseases and their NLRP3 activators are on the right. At Inflazome we believe that inhibition of NLRP3 activation with our molecules will be effective in treating many of these diseases.
Disease
NLRP3 Activators
Cryopyrin Associated Periodic Syndromes (CAPs)
Mutation in the NLRP3 gene
Non-alcoholic steatohepatitis (NASH)
Cholesterol crystals
Alzheimer’s Disease
Aβ plaques
Parkinson’s Disease
α-synuclein
Cardiovascular Disease
Cholesterol crystals
Osteoarthritis
Hydroxyapetite
Gout
Monosodium urate
Pseudogout
Calcium pyrophosphate
Nephrocalcinosis
Calcium salts
Type II diabetes
Free Fatty Acid (FFA), Islet Amyloid Polypeptide (IAPP)
Sjogren’s syndrome
Inflammagenic DNA
Sickle cell disease (SCD)
Heme
Age-related Macular Degeneration (AMD)
Drusen’s deposits, AluRNA
Infections
Virus, fungus and bacteria
Cerebral Malaria
Hemozoin
Asbestosis
Asbestos
Contact hypersensitivity
Nickel
Sunburn
Ultraviolet light
Silicosis
Silica
Cystic fibrosis
Ceramide
Priority Diseases
NLRP3 activation plays a role in many diseases. At Inflazome we have narrowed our focus to develop our suite of therapies for the following diseases where is validated involvement of NLRP3 and the potential to significantly benefit patients:

Imagine a world with effective treatments for Parkinson’s, Alzheimer’s, heart disease and cancer. At Inflazome we are working hard to make this a reality.
Co-founder & Chief Executive Officer
Publications
Details of relevant publications can be found below
Progress Update
Follow our latest clinical advancement and stay up-to-date with our pipeline